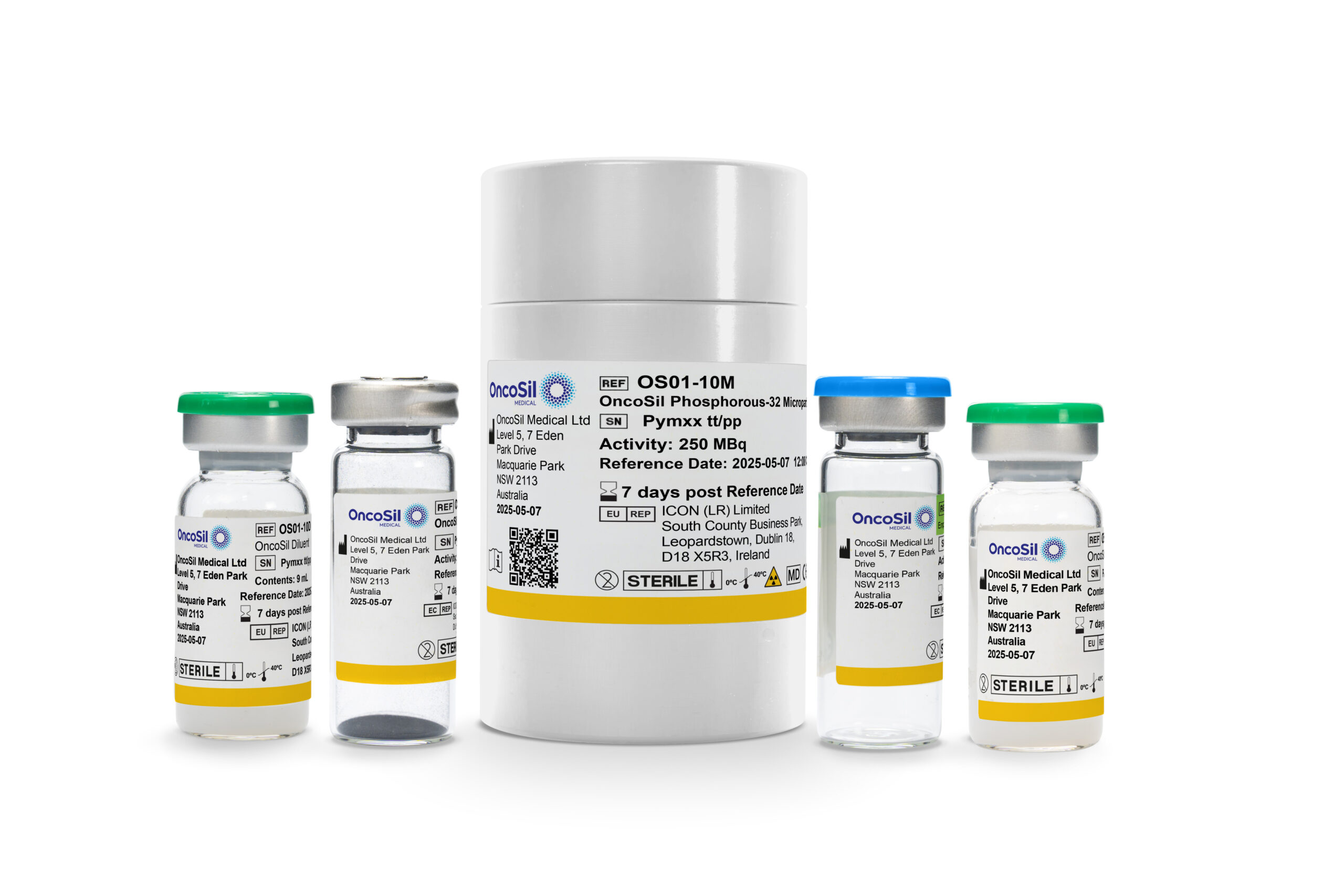
OncoSil™
OncoSil™ is a single-use brachytherapy device used to deliver a pre-determined dose of beta radiation directly into cancerous tissue. It is used in addition to chemotherapy.1



At OncoSil Medical, we are committed to making a positive change through the dedication and compassion of our team. This is why it is important that you get to know the people behind the logo.
We believe in our technology and its ability
to have a truly positive impact in Oncology.
Latest news

OncoSil Medical Receives TGA Approval for OncoSil Device in ...
Read more ...
OSPREY Registry Interim Results Presented at ESGE Days ...
Read more ...
Comparative Analysis Highlights Potential Benefits of OncoSil ...
Read more ...Investor Resources
At OncoSil Medical we have a policy of continuous disclosure. We keep shareholders informed through ASX announcements, the company website, regular newsletters and our annual report. We encourage our shareholders to attend and participate at our annual general meetings.

Annual Report 2025
ASX: OSL
1. The OncoSil™ System is intended to induce prolonged local tumour control and tumour size reduction in patients with locally advanced unresectable pancreatic cancer, in addition to gemcitabine-based chemotherapy, by implantation of radioactive Phosphorous-32 Microparticles into pancreatic tumours under endoscopic ultrasound guidance. OncoSil™ is indicated for the treatment of patients with locally advanced unresectable pancreatic cancer, in addition to gemcitabine-based chemotherapy.
Want to learn more?
OncoSil™ is a brachytherapy device that utilises targeted intratumoural placement of Phosphorous 32 microparticles, delivered via ultrasound-guided endoscopy.


















